Manufacture at Sypharma
Emphasizing efficiency, our production and manufacturing processes consistently deliver high-quality outcomes, ensuring cost-effectiveness alongside flexible capacity and scalability.



Our Agile
Facility
GMP Compliant Manufacturing
Sypharma operates state-of-the-art Good Manufacturing Practice (cGMP) facilities compliant with multiple regulatory standards.
Our licenses encompass TGA (Therapeutic Goods Administration) for medicines and medical devices, and APVMA (Australian Pesticide and Veterinary Medicines Authority) accreditation.

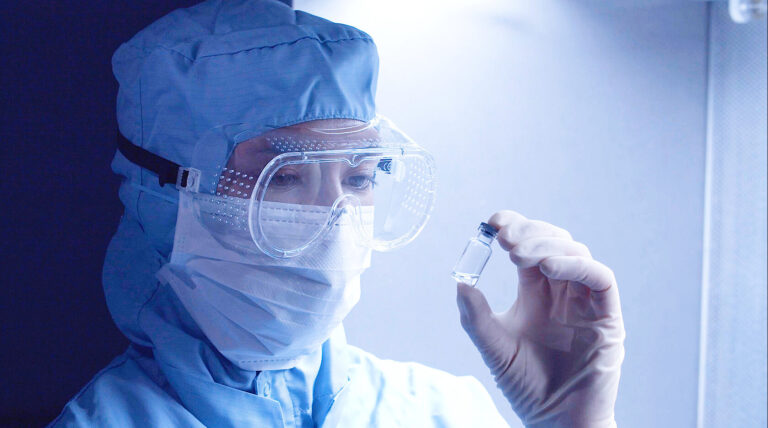
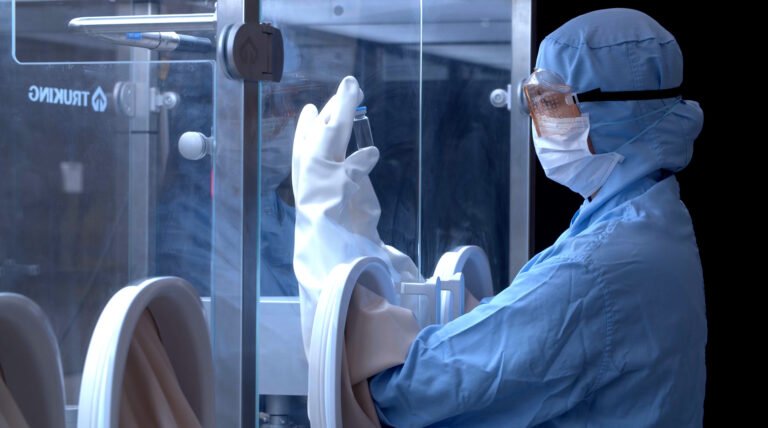
Single Use Manufacturing
Efficiency, Flexibility and Quality
- Reduced Contamination Risk: minimize the risk of cross-contamination between batches, ensuring product purity and safety.
- Increased Flexibility: allows for greater flexibility in manufacturing processes, facilitating rapid changeovers between different products or formulations.
- Cost Savings: eliminating the need for cleaning and sterilization of traditional equipment reduces operational costs associated with labor, utilities, and downtime.
- Scalability: easier scaling of production capacity, allowing companies to adjust manufacturing volumes more efficiently in response to market demand.
- Faster Time to Market: simplified setup and validation processes, accelerating the development and production of pharmaceutical products, reducing time to market.


Single Use
Manufacturing
Efficiency, Flexibility and Quality
Advanced single-use manufacturing solutions for clinical trial injectables, ensuring flexible, efficient, and contamination-free GMP production
Reduced Contamination Risk
Minimize the risk of cross-contamination between batches, ensuring product purity and safety.
Increased Flexibility
Allows for greater flexibility in manufacturing processes, facilitating rapid changeovers between different products or formulations.
Cost Savings
Eliminating the need for cleaning and sterilization of traditional equipment reduces operational costs associated with labor, utilities, and downtime.
Scalability
Easier scaling of production capacity, allowing companies to adjust manufacturing volumes more efficiently in response to market demand.
Faster Time to Market
Simplified setup and validation processes, accelerating the development and production of pharmaceutical products, reducing time to market.






Intravenous Fluid
Manufacturing
Automation, Optimization and Digitalization
- Precision and Accuracy: ensured precise measurement and mixing of IV solutions, reducing the risk of errors in formulation.
- Consistency: guaranteed uniformity in product quality and composition, enhancing patient safety and treatment efficacy.
- Increased Efficiency: reduced manual intervention and streamlines production workflows, leading to higher output rates and faster turnaround times.
- Cost Reduction: minimized labor requirements and optimized resource utilization, with lower operational costs.


Intravenous Fluid Manufacture
Automation, Optimization and Digitalization
Specialising in the GMP manufacture of intravenous fluids for clinical and commercial use, delivering precision, sterility, and quality at scale.
Precision and Accuracy
Ensured precise measurement and mixing of IV solutions, reducing the risk of errors in formulation.
Consistency
Guaranteed uniformity in product quality and composition, enhancing patient safety and treatment efficacy.
Increased Efficiency
Reduced manual intervention and streamlines production workflows, leading to higher output rates and faster turnaround times.
Cost Reduction
Minimized labor requirements and optimized resource utilization, with lower operational costs.






Non-sterile
Manufacture
Customization, Adaption and Solution
- Customer Consultation: The manufacturing process begins with a thorough consultation with the customer to understand their needs, preferences, and specifications for the desired liquid product.
- Formulation Development: Based on the customer’s requirements, formulation experts develop a customized recipe or formulation for the liquid product.
- Prototype Production: Once the formulation is developed, prototypes of the liquid product are produced for testing and evaluation.
- Scale-Up and Production: Once the formulation has been finalized and approved by the customer, it is scaled up for commercial production.
- Quality Assurance: Throughout the manufacturing process, rigorous quality assurance measures are implemented to ensure the integrity, purity, and safety of the liquid product.


Non-sterile
Manufacture
Customization, Adaption and Solution
Sypharma manufactures GMP-grade excipients to support pharmaceutical and biopharmaceutical formulation with consistent quality and regulatory compliance.
Customer Consultation
The manufacturing process begins with a thorough consultation with the customer to understand their needs, preferences, and specifications for the desired liquid product.
Formulation Development
Based on the customer’s requirements, formulation experts develop a customized recipe or formulation for the liquid product.
Prototype Production
Once the formulation is developed, prototypes of the liquid product are produced for testing and evaluation.
Scale-Up and Production
Once the formulation has been finalized and approved by the customer, it is scaled up for commercial production.
Quality Assurance
Throughout the manufacturing process, rigorous quality assurance measures are implemented to ensure the integrity, purity, and safety of the liquid product.





In-house
Quality Control
Accuracy, Compliance and Reliability
In-house Chemistry & Microbiology laboratories enhances our ability to maintain stringent quality standards, support product development initiatives, ensure regulatory compliance, and ultimately deliver high-quality pharmaceutical products to our customers.
Rapid Testing and Analysis
Allowing for quick testing and analysis of raw materials, in-process samples, and finished products.
Quality Control Assurance
Implementing robust quality control measures throughout the manufacturing process.
Enhanced Product Development
Evaluating new formulations, assess product stability, and optimize manufacturing processes, leading to the continuous improvement of your product offerings.
Cost Efficiency
Minimizing the need for outsourcing these services to third-party laboratories, resulting in cost savings and greater control over expenses.




Our dedication to quality is uncompromising.
And it covers every stage of everything we do. From development, clinical testing, and production through to commercialisation. Hundreds of organisations around the world trust Sypharma to fast track drug development through every stage of research, development, quality, safety and commercialisation.
Unlocking resources
Collaboration & Potential
We work together with innovative biotech developers to your drug production destination means the world to us, and our dedicated team of experts will support and guide you from the earliest stage of development through to commercialization.













